Embedding iron oxide into liposome bilayer to trigger ferroptosis
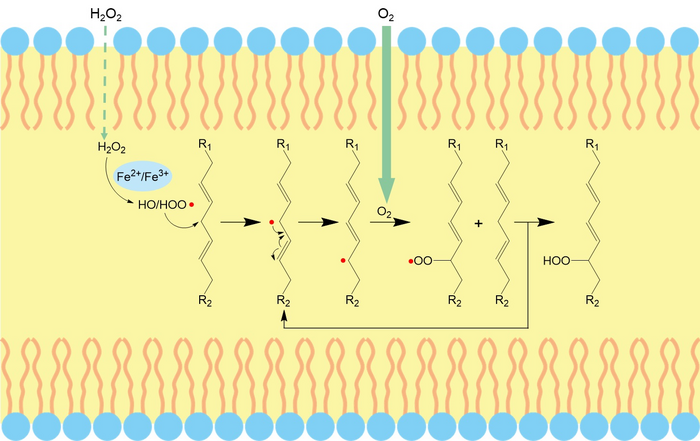
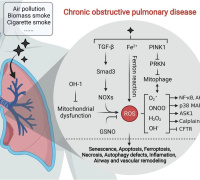
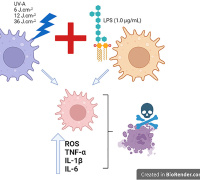
Ferroptosis, an iron-dependent regulated cell death process driven by excessive lipid peroxides and membrane injury, can enhance cancer vulnerability to chemotherapy. Lipid peroxidation of unsaturated lipids (UL) in biological membranes is a key to inducing ferroptosis. However, there is a significant thermodynamic barrier for hydrophilic polar nonelectrolytes (e.g., hydrogen peroxide (H2O2) and hydroxyl radicals (•OH)) and ions to diffuse toward the center of the lipid bilayer for the initiation of lipid peroxidation. Improving the local content of diffusion-limited ROS in the lipid bilayer is a potential strategy to initiate peroxidation by inducing ferroptosis.
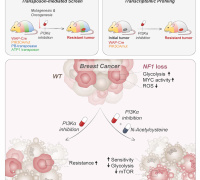
A research team at the Institute of Chemistry, Chinese Academy of Sciences, published an online article in the National Science Review (NSR) entitled ‘Liposomes embedded with PEGylated iron oxide nanoparticles enabling fertility and combination in cancer’. Yang Liu, a doctoral student at the Institute of Chemistry, Chinese Academy of Sciences, and Xuebo Quan, a postdoctoral fellow at Shenzhen Bay Laboratory, are co-first authors. Chunli Bai, an academician (CAS), Wang Chunru, a researcher, and Li Jie, an assistant researcher at the Institute of Chemistry, Chinese Academy of Sciences, are corresponding authors.
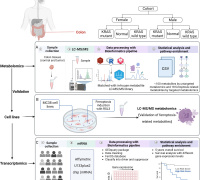
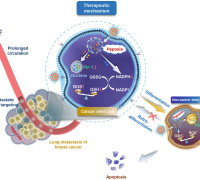
The researchers embedded PEGylated ultra-small γ-Fe2O3 nanoparticles (IO-PEG) into the bilayer of liposomes to construct Lp-IO liposomes. In the lipid bilayer, IO-PEG promotes the intrabilayer generation of •OH from H2O2. And the intrabilayer UL was peroxidized rapidly to LPO by •OH. At the same time, molecular dynamics simulation showed that the integration of amphiphilic PEG moieties with liposomal membranes improved its permeability to H2O2 and •OH, further promoting the production of LPO. Liquid chromatography-mass spectrometry analysis showed that unsaturated PC, LPC, and SM were oxygenated in Lp-IO, and the peroxidation vulnerability of PC/LPC/SM was generally enhanced with their degree of unsaturation.
Lp-IO significantly improved ROS and LPO levels in vitro for inducing tumor cell ferroptosis. Neither liposome prototype (Lp) nor IO-PEG caused apparent inhibition in tumor cells. Further, the researchers embedded IO-PEG into the bilayer of liposomes consisting of 16:0 PC to construct UL-free Lp-IO liposomes and discovered that UL-free Lp-IO could not induce ferroptosis. Thus, unsaturated lipids of the lipid bilayer play a critical role in the Lp-IO system to initiate intrabilayer lipid peroxidation and induce tumor cell ferroptosis.
In vivo, Lp-IO had a tumor inhibition rate of ~66.2% and negligible side effects at a 2.5 mg Fe/kg dose. Besides, Lp-IO enabled traceable magnetic resonance imaging and pH/ROS dual-responsive drug delivery. Synergistic antineoplastic effects of chemotherapy and ferroptosis were achieved by delivering doxorubicin (capable of xCT and GPX-4 inhibition) with Lp-IO.
This study revealed the critical role of intrabilayer lipid peroxidation in inducing cell ferroptosis and provided an effective strategy to initiate lipid peroxidation for tumor cell ferroptosis. It is also expected to achieve effective treatment of ferroptosis-related diseases through precise regulation of ferroptosis.
News source: www.eurekalert.org
Image Credit: ©Science China Press
Media Contact:
Redox Medicine Society
This email address is being protected from spambots. You need JavaScript enabled to view it.
Redox Medicine 2023 Congress
June 21-23, 2023 - Paris, France
Website | LinkedIn | Facebook